3. 结构
3.1 二维结构
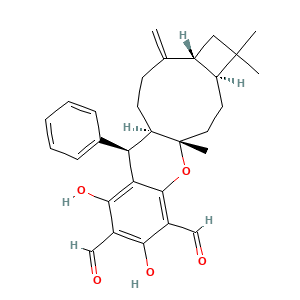
3.2 三维结构
-1
-2
-3
69 73 0 1 0 0 0 0 0999 V2000
-0.5816 0.6957 -1.7417 O 0 0 0 0 0 0 0 0 0 0 0 0
-2.5667 -0.5771 2.4570 O 0 0 0 0 0 0 0 0 0 0 0 0
-4.1005 3.2806 0.1020 O 0 0 0 0 0 0 0 0 0 0 0 0
-3.2289 3.4665 -2.3988 O 0 0 0 0 0 0 0 0 0 0 0 0
-5.1087 2.5460 2.3071 O 0 0 0 0 0 0 0 0 0 0 0 0
3.7478 1.0489 0.0334 C 0 0 2 0 0 0 0 0 0 0 0 0
5.2507 1.4649 0.0948 C 0 0 0 0 0 0 0 0 0 0 0 0
4.2030 -0.2516 0.7900 C 0 0 1 0 0 0 0 0 0 0 0 0
5.3852 0.5802 1.3485 C 0 0 0 0 0 0 0 0 0 0 0 0
3.2216 0.9598 -1.4045 C 0 0 0 0 0 0 0 0 0 0 0 0
0.6937 0.0388 -1.5001 C 0 0 1 0 0 0 0 0 0 0 0 0
1.7143 1.2267 -1.5321 C 0 0 0 0 0 0 0 0 0 0 0 0
0.7732 -0.7436 -0.1481 C 0 0 2 0 0 0 0 0 0 0 0 0
3.2108 -0.8609 1.7292 C 0 0 0 0 0 0 0 0 0 0 0 0
6.0632 0.9725 -1.0826 C 0 0 0 0 0 0 0 0 0 0 0 0
5.5823 2.9184 0.3787 C 0 0 0 0 0 0 0 0 0 0 0 0
1.8196 -1.8863 -0.1265 C 0 0 0 0 0 0 0 0 0 0 0 0
2.4776 -2.0939 1.2456 C 0 0 0 0 0 0 0 0 0 0 0 0
-0.5971 -1.1667 0.4525 C 0 0 1 0 0 0 0 0 0 0 0 0
0.9015 -0.8456 -2.7428 C 0 0 0 0 0 0 0 0 0 0 0 0
-1.5130 0.0306 0.3888 C 0 0 0 0 0 0 0 0 0 0 0 0
2.9362 -0.3167 2.9244 C 0 0 0 0 0 0 0 0 0 0 0 0
-1.4349 0.8981 -0.6993 C 0 0 0 0 0 0 0 0 0 0 0 0
-1.2361 -2.4442 -0.0740 C 0 0 0 0 0 0 0 0 0 0 0 0
-2.4668 0.2679 1.3886 C 0 0 0 0 0 0 0 0 0 0 0 0
-2.3108 1.9804 -0.8030 C 0 0 0 0 0 0 0 0 0 0 0 0
-2.0993 -2.3922 -1.1684 C 0 0 0 0 0 0 0 0 0 0 0 0
-0.9584 -3.6666 0.5377 C 0 0 0 0 0 0 0 0 0 0 0 0
-3.3281 1.3611 1.2958 C 0 0 0 0 0 0 0 0 0 0 0 0
-3.2509 2.2165 0.1990 C 0 0 0 0 0 0 0 0 0 0 0 0
-2.2451 2.8733 -1.9564 C 0 0 0 0 0 0 0 0 0 0 0 0
-2.6850 -3.5626 -1.6512 C 0 0 0 0 0 0 0 0 0 0 0 0
-1.5439 -4.8369 0.0550 C 0 0 0 0 0 0 0 0 0 0 0 0
-2.4073 -4.7850 -1.0394 C 0 0 0 0 0 0 0 0 0 0 0 0
-4.3176 1.6030 2.3406 C 0 0 0 0 0 0 0 0 0 0 0 0
3.1298 1.7168 0.6520 H 0 0 0 0 0 0 0 0 0 0 0 0
4.5820 -1.0335 0.1154 H 0 0 0 0 0 0 0 0 0 0 0 0
5.2124 1.1169 2.2871 H 0 0 0 0 0 0 0 0 0 0 0 0
6.3283 0.0327 1.4504 H 0 0 0 0 0 0 0 0 0 0 0 0
3.5175 0.0314 -1.8990 H 0 0 0 0 0 0 0 0 0 0 0 0
3.6867 1.7695 -1.9823 H 0 0 0 0 0 0 0 0 0 0 0 0
1.5620 1.7415 -2.4935 H 0 0 0 0 0 0 0 0 0 0 0 0
1.4097 1.9747 -0.7868 H 0 0 0 0 0 0 0 0 0 0 0 0
1.1189 0.0225 0.5607 H 0 0 0 0 0 0 0 0 0 0 0 0
5.9029 1.5855 -1.9765 H 0 0 0 0 0 0 0 0 0 0 0 0
5.8645 -0.0681 -1.3570 H 0 0 0 0 0 0 0 0 0 0 0 0
7.1354 1.0301 -0.8575 H 0 0 0 0 0 0 0 0 0 0 0 0
5.3411 3.5513 -0.4824 H 0 0 0 0 0 0 0 0 0 0 0 0
6.6459 3.0481 0.6068 H 0 0 0 0 0 0 0 0 0 0 0 0
5.0116 3.3015 1.2320 H 0 0 0 0 0 0 0 0 0 0 0 0
1.3872 -2.8303 -0.4704 H 0 0 0 0 0 0 0 0 0 0 0 0
2.6207 -1.6879 -0.8413 H 0 0 0 0 0 0 0 0 0 0 0 0
1.7225 -2.4122 1.9731 H 0 0 0 0 0 0 0 0 0 0 0 0
3.1980 -2.9181 1.1706 H 0 0 0 0 0 0 0 0 0 0 0 0
-0.3753 -1.3436 1.5149 H 0 0 0 0 0 0 0 0 0 0 0 0
0.7221 -0.2711 -3.6599 H 0 0 0 0 0 0 0 0 0 0 0 0
1.9039 -1.2725 -2.8156 H 0 0 0 0 0 0 0 0 0 0 0 0
0.1810 -1.6688 -2.7596 H 0 0 0 0 0 0 0 0 0 0 0 0
3.4090 0.5872 3.2866 H 0 0 0 0 0 0 0 0 0 0 0 0
2.1963 -0.7647 3.5808 H 0 0 0 0 0 0 0 0 0 0 0 0
-2.3345 -1.4679 -1.6838 H 0 0 0 0 0 0 0 0 0 0 0 0
-0.2944 -3.7244 1.3953 H 0 0 0 0 0 0 0 0 0 0 0 0
-1.2703 2.9638 -2.4643 H 0 0 0 0 0 0 0 0 0 0 0 0
-3.3549 -3.5230 -2.5052 H 0 0 0 0 0 0 0 0 0 0 0 0
-1.3288 -5.7886 0.5321 H 0 0 0 0 0 0 0 0 0 0 0 0
-2.2206 -1.4547 2.2333 H 0 0 0 0 0 0 0 0 0 0 0 0
-2.8629 -5.6963 -1.4154 H 0 0 0 0 0 0 0 0 0 0 0 0
-4.3442 0.9015 3.1904 H 0 0 0 0 0 0 0 0 0 0 0 0
-3.8493 3.8630 -0.6316 H 0 0 0 0 0 0 0 0 0 0 0 0
1 11 1 0 0 0 0
1 23 1 0 0 0 0
2 25 1 0 0 0 0
2 66 1 0 0 0 0
3 30 1 0 0 0 0
3 69 1 0 0 0 0
4 31 2 0 0 0 0
5 35 2 0 0 0 0
6 7 1 0 0 0 0
6 8 1 0 0 0 0
6 10 1 0 0 0 0
6 36 1 0 0 0 0
7 9 1 0 0 0 0
7 15 1 0 0 0 0
7 16 1 0 0 0 0
8 9 1 0 0 0 0
8 14 1 0 0 0 0
8 37 1 0 0 0 0
9 38 1 0 0 0 0
9 39 1 0 0 0 0
10 12 1 0 0 0 0
10 40 1 0 0 0 0
10 41 1 0 0 0 0
11 12 1 0 0 0 0
11 13 1 0 0 0 0
11 20 1 0 0 0 0
12 42 1 0 0 0 0
12 43 1 0 0 0 0
13 17 1 0 0 0 0
13 19 1 0 0 0 0
13 44 1 0 0 0 0
14 18 1 0 0 0 0
14 22 2 0 0 0 0
15 45 1 0 0 0 0
15 46 1 0 0 0 0
15 47 1 0 0 0 0
16 48 1 0 0 0 0
16 49 1 0 0 0 0
16 50 1 0 0 0 0
17 18 1 0 0 0 0
17 51 1 0 0 0 0
17 52 1 0 0 0 0
18 53 1 0 0 0 0
18 54 1 0 0 0 0
19 21 1 0 0 0 0
19 24 1 0 0 0 0
19 55 1 0 0 0 0
20 56 1 0 0 0 0
20 57 1 0 0 0 0
20 58 1 0 0 0 0
21 23 1 0 0 0 0
21 25 2 0 0 0 0
22 59 1 0 0 0 0
22 60 1 0 0 0 0
23 26 2 0 0 0 0
24 27 2 0 0 0 0
24 28 1 0 0 0 0
25 29 1 0 0 0 0
26 30 1 0 0 0 0
26 31 1 0 0 0 0
27 32 1 0 0 0 0
27 61 1 0 0 0 0
28 33 2 0 0 0 0
28 62 1 0 0 0 0
29 30 2 0 0 0 0
29 35 1 0 0 0 0
31 63 1 0 0 0 0
32 34 2 0 0 0 0
32 64 1 0 0 0 0
33 34 1 0 0 0 0
33 65 1 0 0 0 0
34 67 1 0 0 0 0
35 68 1 0 0 0 0
4. 国际命名与标识
4.1 IUPAC Name
(1S,4S,7R,11R,12R)-14,16-dihydroxy-1,5,5-trimethyl-8-methylidene-12-phenyl-19-oxatetracyclo[9.8.0.04,7.013,18]nonadeca-13,15,17-triene-15,17-dicarbaldehyde
4.2 InChl
InChI=1S/C30H34O5/c1-17-10-11-23-24(18-8-6-5-7-9-18)25-27(34)20(15-31)26(33)21(16-32)28(25)35-30(23,4)13-12-22-19(17)14-29(22,2)3/h5-9,15-16,19,22-24,33-34H,1,10-14H2,2-4H3/t19-,22-,23+,24-,30-/m0/s1
4.3 InChlKey
NSFVENNIBGTQJE-QIBYHJKFSA-N
4.4 Canonical SMILES
CC1(CC2C1CCC3(C(CCC2=C)C(C4=C(C(=C(C(=C4O3)C=O)O)C=O)O)C5=CC=CC=C5)C)C
4.5 lsomeric SMILES
C[C@]12CC[C@H]3[C@@H](CC3(C)C)C(=C)CC[C@@H]1[C@@H](C4=C(C(=C(C(=C4O2)C=O)O)C=O)O)C5=CC=CC=C5
4.6 SDF文件
5. 波谱数据
5.1 13C核磁共振谱(13C NMR)
5.2 1H核磁共振谱(1H NMR)
5.3 质谱(MS)
5.4 红外光谱(IR)
5.5 紫外/可见光谱(UV/Vis)
6. 相关药材
7. 相关靶点
8. 相关疾病