3. 结构
3.1 二维结构
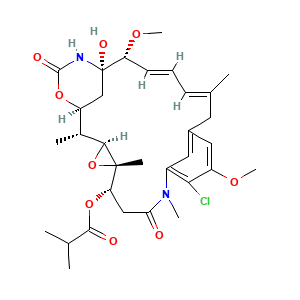
3.2 三维结构
-1
-2
-3
87 90 0 1 0 0 0 0 0999 V2000
-6.2658 -1.6247 -1.1959 Cl 0 0 0 0 0 0 0 0 0 0 0 0
0.9463 -3.2767 -1.8866 O 0 0 0 0 0 0 0 0 0 0 0 0
0.4290 -2.2851 1.3875 O 0 0 0 0 0 0 0 0 0 0 0 0
5.1918 -0.8500 -1.6630 O 0 0 0 0 0 0 0 0 0 0 0 0
4.1987 0.5325 1.4314 O 0 0 0 0 0 0 0 0 0 0 0 0
4.1658 3.3046 1.0556 O 0 0 0 0 0 0 0 0 0 0 0 0
-2.3756 -3.1736 1.8175 O 0 0 0 0 0 0 0 0 0 0 0 0
7.0782 0.3849 -1.7539 O 0 0 0 0 0 0 0 0 0 0 0 0
0.6462 -0.0649 0.8018 O 0 0 0 0 0 0 0 0 0 0 0 0
-6.6887 1.1694 -1.9374 O 0 0 0 0 0 0 0 0 0 0 0 0
5.3494 1.1659 -0.4757 N 0 0 0 0 0 0 0 0 0 0 0 0
-3.9227 -1.7645 0.8075 N 0 0 0 0 0 0 0 0 0 0 0 0
0.6733 -2.1950 -0.9865 C 0 0 2 0 0 0 0 0 0 0 0 0
2.0445 -2.8240 -1.0813 C 0 0 2 0 0 0 0 0 0 0 0 0
3.1633 -2.1855 -1.8636 C 0 0 1 0 0 0 0 0 0 0 0 0
-0.2418 -2.5462 0.1512 C 0 0 2 0 0 0 0 0 0 0 0 0
3.9582 -1.1871 -0.9827 C 0 0 2 0 0 0 0 0 0 0 0 0
0.3893 -0.9388 -1.7497 C 0 0 0 0 0 0 0 0 0 0 0 0
4.0733 -3.2984 -2.4094 C 0 0 0 0 0 0 0 0 0 0 0 0
3.1674 0.0918 -0.7049 C 0 0 0 0 0 0 0 0 0 0 0 0
-1.5132 -1.6862 0.1375 C 0 0 0 0 0 0 0 0 0 0 0 0
4.0204 1.0607 0.1147 C 0 0 1 0 0 0 0 0 0 0 0 0
3.3794 2.4680 0.2018 C 0 0 1 0 0 0 0 0 0 0 0 0
-2.6250 -2.2625 1.0223 C 0 0 0 0 0 0 0 0 0 0 0 0
5.9400 0.2386 -1.3105 C 0 0 0 0 0 0 0 0 0 0 0 0
0.8212 -0.9907 1.5833 C 0 0 0 0 0 0 0 0 0 0 0 0
1.5252 -0.8726 2.9238 C 0 0 0 0 0 0 0 0 0 0 0 0
1.9658 2.4720 0.7264 C 0 0 0 0 0 0 0 0 0 0 0 0
-4.2260 -0.4744 0.2626 C 0 0 0 0 0 0 0 0 0 0 0 0
-5.0505 -2.6310 1.1686 C 0 0 0 0 0 0 0 0 0 0 0 0
0.5119 -0.5259 4.0082 C 0 0 0 0 0 0 0 0 0 0 0 0
2.2669 -2.1634 3.2612 C 0 0 0 0 0 0 0 0 0 0 0 0
0.9082 2.8253 -0.0218 C 0 0 0 0 0 0 0 0 0 0 0 0
3.6704 4.6329 1.0688 C 0 0 0 0 0 0 0 0 0 0 0 0
-3.4701 0.6521 0.6198 C 0 0 0 0 0 0 0 0 0 0 0 0
-5.3357 -0.2891 -0.5739 C 0 0 0 0 0 0 0 0 0 0 0 0
-0.4491 2.8380 0.4794 C 0 0 0 0 0 0 0 0 0 0 0 0
-3.7668 1.9272 0.1116 C 0 0 0 0 0 0 0 0 0 0 0 0
-2.9156 3.1125 0.5010 C 0 0 0 0 0 0 0 0 0 0 0 0
-1.5708 3.1438 -0.2050 C 0 0 0 0 0 0 0 0 0 0 0 0
-5.6387 0.9743 -1.0872 C 0 0 0 0 0 0 0 0 0 0 0 0
-4.8535 2.0764 -0.7546 C 0 0 0 0 0 0 0 0 0 0 0 0
-1.6273 3.5578 -1.6527 C 0 0 0 0 0 0 0 0 0 0 0 0
-7.9475 1.4819 -1.3472 C 0 0 0 0 0 0 0 0 0 0 0 0
2.3821 -3.4650 -0.2769 H 0 0 0 0 0 0 0 0 0 0 0 0
2.8067 -1.6698 -2.7608 H 0 0 0 0 0 0 0 0 0 0 0 0
-0.4876 -3.6141 0.1303 H 0 0 0 0 0 0 0 0 0 0 0 0
4.2360 -1.6866 -0.0460 H 0 0 0 0 0 0 0 0 0 0 0 0
0.0531 -0.0612 -1.2047 H 0 0 0 0 0 0 0 0 0 0 0 0
1.1925 -0.6008 -2.4014 H 0 0 0 0 0 0 0 0 0 0 0 0
-0.4296 -1.1543 -2.4511 H 0 0 0 0 0 0 0 0 0 0 0 0
3.4918 -4.0286 -2.9833 H 0 0 0 0 0 0 0 0 0 0 0 0
4.8445 -2.8977 -3.0749 H 0 0 0 0 0 0 0 0 0 0 0 0
4.5691 -3.8379 -1.5948 H 0 0 0 0 0 0 0 0 0 0 0 0
2.8978 0.5705 -1.6531 H 0 0 0 0 0 0 0 0 0 0 0 0
2.3075 -0.1944 -0.1682 H 0 0 0 0 0 0 0 0 0 0 0 0
-1.9341 -1.6929 -0.8764 H 0 0 0 0 0 0 0 0 0 0 0 0
-1.3627 -0.6574 0.4274 H 0 0 0 0 0 0 0 0 0 0 0 0
3.4057 2.9037 -0.8079 H 0 0 0 0 0 0 0 0 0 0 0 0
5.9240 1.9509 -0.1842 H 0 0 0 0 0 0 0 0 0 0 0 0
4.9257 -0.1107 1.3867 H 0 0 0 0 0 0 0 0 0 0 0 0
2.2561 -0.0643 2.8454 H 0 0 0 0 0 0 0 0 0 0 0 0
1.8267 2.1895 1.7676 H 0 0 0 0 0 0 0 0 0 0 0 0
-5.1912 -3.4000 0.4042 H 0 0 0 0 0 0 0 0 0 0 0 0
-5.9560 -2.0659 1.4072 H 0 0 0 0 0 0 0 0 0 0 0 0
-4.7919 -3.1723 2.0869 H 0 0 0 0 0 0 0 0 0 0 0 0
1.0035 -0.4017 4.9783 H 0 0 0 0 0 0 0 0 0 0 0 0
-0.2506 -1.3067 4.1065 H 0 0 0 0 0 0 0 0 0 0 0 0
-0.0071 0.4085 3.7673 H 0 0 0 0 0 0 0 0 0 0 0 0
2.8893 -2.0205 4.1513 H 0 0 0 0 0 0 0 0 0 0 0 0
2.9256 -2.4656 2.4394 H 0 0 0 0 0 0 0 0 0 0 0 0
1.5835 -2.9936 3.4716 H 0 0 0 0 0 0 0 0 0 0 0 0
1.0781 3.1044 -1.0558 H 0 0 0 0 0 0 0 0 0 0 0 0
2.7634 4.7205 1.6743 H 0 0 0 0 0 0 0 0 0 0 0 0
3.5000 5.0137 0.0562 H 0 0 0 0 0 0 0 0 0 0 0 0
4.4307 5.2651 1.5378 H 0 0 0 0 0 0 0 0 0 0 0 0
-2.6754 0.5695 1.3577 H 0 0 0 0 0 0 0 0 0 0 0 0
-0.5521 2.5602 1.5281 H 0 0 0 0 0 0 0 0 0 0 0 0
-3.4525 4.0482 0.2967 H 0 0 0 0 0 0 0 0 0 0 0 0
-2.7882 3.0994 1.5921 H 0 0 0 0 0 0 0 0 0 0 0 0
-5.0932 3.0544 -1.1650 H 0 0 0 0 0 0 0 0 0 0 0 0
-2.1703 4.5038 -1.7521 H 0 0 0 0 0 0 0 0 0 0 0 0
-2.1472 2.7981 -2.2453 H 0 0 0 0 0 0 0 0 0 0 0 0
-0.6457 3.7093 -2.1059 H 0 0 0 0 0 0 0 0 0 0 0 0
-8.6967 1.5281 -2.1424 H 0 0 0 0 0 0 0 0 0 0 0 0
-7.9028 2.4574 -0.8528 H 0 0 0 0 0 0 0 0 0 0 0 0
-8.2481 0.7132 -0.6284 H 0 0 0 0 0 0 0 0 0 0 0 0
1 36 1 0 0 0 0
2 13 1 0 0 0 0
2 14 1 0 0 0 0
3 16 1 0 0 0 0
3 26 1 0 0 0 0
4 17 1 0 0 0 0
4 25 1 0 0 0 0
5 22 1 0 0 0 0
5 61 1 0 0 0 0
6 23 1 0 0 0 0
6 34 1 0 0 0 0
7 24 2 0 0 0 0
8 25 2 0 0 0 0
9 26 2 0 0 0 0
10 41 1 0 0 0 0
10 44 1 0 0 0 0
11 22 1 0 0 0 0
11 25 1 0 0 0 0
11 60 1 0 0 0 0
12 24 1 0 0 0 0
12 29 1 0 0 0 0
12 30 1 0 0 0 0
13 14 1 0 0 0 0
13 16 1 0 0 0 0
13 18 1 0 0 0 0
14 15 1 0 0 0 0
14 45 1 0 0 0 0
15 17 1 0 0 0 0
15 19 1 0 0 0 0
15 46 1 0 0 0 0
16 21 1 0 0 0 0
16 47 1 0 0 0 0
17 20 1 0 0 0 0
17 48 1 0 0 0 0
18 49 1 0 0 0 0
18 50 1 0 0 0 0
18 51 1 0 0 0 0
19 52 1 0 0 0 0
19 53 1 0 0 0 0
19 54 1 0 0 0 0
20 22 1 0 0 0 0
20 55 1 0 0 0 0
20 56 1 0 0 0 0
21 24 1 0 0 0 0
21 57 1 0 0 0 0
21 58 1 0 0 0 0
22 23 1 0 0 0 0
23 28 1 0 0 0 0
23 59 1 0 0 0 0
26 27 1 0 0 0 0
27 31 1 0 0 0 0
27 32 1 0 0 0 0
27 62 1 0 0 0 0
28 33 2 0 0 0 0
28 63 1 0 0 0 0
29 35 1 0 0 0 0
29 36 2 0 0 0 0
30 64 1 0 0 0 0
30 65 1 0 0 0 0
30 66 1 0 0 0 0
31 67 1 0 0 0 0
31 68 1 0 0 0 0
31 69 1 0 0 0 0
32 70 1 0 0 0 0
32 71 1 0 0 0 0
32 72 1 0 0 0 0
33 37 1 0 0 0 0
33 73 1 0 0 0 0
34 74 1 0 0 0 0
34 75 1 0 0 0 0
34 76 1 0 0 0 0
35 38 2 0 0 0 0
35 77 1 0 0 0 0
36 41 1 0 0 0 0
37 40 2 0 0 0 0
37 78 1 0 0 0 0
38 39 1 0 0 0 0
38 42 1 0 0 0 0
39 40 1 0 0 0 0
39 79 1 0 0 0 0
39 80 1 0 0 0 0
40 43 1 0 0 0 0
41 42 2 0 0 0 0
42 81 1 0 0 0 0
43 82 1 0 0 0 0
43 83 1 0 0 0 0
43 84 1 0 0 0 0
44 85 1 0 0 0 0
44 86 1 0 0 0 0
44 87 1 0 0 0 0
4. 国际命名与标识
4.1 IUPAC Name
[(1S,2R,3S,5S,6S,16E,18E,20R,21S)-11-chloro-21-hydroxy-12,20-dimethoxy-2,5,9,16-tetramethyl-8,23-dioxo-4,24-dioxa-9,22-diazatetracyclo[19.3.1.110,14.03,5]hexacosa-10,12,14(26),16,18-pentaen-6-yl] 2-methylpropanoate
4.2 InChl
InChI=1S/C32H43ClN2O9/c1-17(2)29(37)43-25-15-26(36)35(6)21-13-20(14-22(40-7)27(21)33)12-18(3)10-9-11-24(41-8)32(39)16-23(42-30(38)34-32)19(4)28-31(25,5)44-28/h9-11,13-14,17,19,23-25,28,39H,12,15-16H2,1-8H3,(H,34,38)/b11-9+,18-10+/t19-,23+,24-,25+,28+,31+,32+/m1/s1
4.3 InChlKey
OPQNCARIZFLNLF-JBHFWYGFSA-N
4.4 Canonical SMILES
CC1C2CC(C(C=CC=C(CC3=CC(=C(C(=C3)OC)Cl)N(C(=O)CC(C4(C1O4)C)OC(=O)C(C)C)C)C)OC)(NC(=O)O2)O
4.5 lsomeric SMILES
C[C@@H]1[C@@H]2C[C@]([C@@H](/C=C/C=C(/CC3=CC(=C(C(=C3)OC)Cl)N(C(=O)C[C@@H]([C@]4([C@H]1O4)C)OC(=O)C(C)C)C)\C)OC)(NC(=O)O2)O
4.6 SDF文件
5. 波谱数据
5.1 13C核磁共振谱(13C NMR)
5.2 1H核磁共振谱(1H NMR)
5.3 质谱(MS)
5.4 红外光谱(IR)
5.5 紫外/可见光谱(UV/Vis)
6. 相关药材
7. 相关靶点
8. 相关疾病